Cdcl3 Nmr Solvent Peak

Cdcl3 Nmr Solvent Peak. Click on the arrow by to see the different options. The chemical shifts were read and are presented in table 1. Here, the chemical shifts of tms and the proton and deuterium chemical shifts of t … Common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist gregory r. 3.8, gives rise to eight signals at 77.91, 110.12, 115.04, 118.85, 122.89, 132.25, 138.81, and 182.85 ppm. The standard for chemical shift is dilute tetramethylsilane (tms) in cdcl3, but many measurements are made relative to tms in other solvents, the proton resonance of the solvent peak or relative to the lock frequency. Why does dmso show quintet in 1h nmr? Furthermore, 13 c decoupling is useful, when solvent 13 c satellite signals overlap with sample signals. Whenever you run a 13c spectrum in cdcl₃, you always get a triplet solvent peak at 77.5 ppm.
A) residual h 2 o and hod in the d 2 o bottle; The gui that pops upallows you to annotate the peak with any text. Molecular weight, density (25 °c), melting point (mp), and boiling point (bp) are also given. Gottlieb, 3 abraham nudelman, brian m.
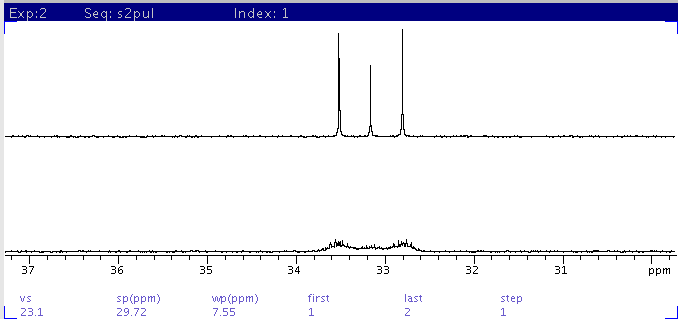
A common solvent for dissolving compounds for 1h and 13c nmr spectroscopy is deuteriochloroform, dccl3.
Find the unknown molecule (the peaks at around 7.3ppm in the 1h nmr are due to the cdcl3 solvent, as are the three small peaks at around 77ppm in the 13c nmr) this problem has been solved! What is the reason behind it? However, deuteration is not 100%, so signals for the residual protons are observed. Common spectroscopic solvents are available commercially in different degrees of deuteration. The chemical shifts were read and are presented in table 1. Why does the water peak appear at different chemical shift values (ppm) in different solvents in proton nmr spectra? Now let us consider that you are doing 1hnmr, and as for nmr you need to first dissolve your compound in a solvent and for this you need to check weather. Here, the chemical shifts of tms and the proton and deuterium chemical shifts of t … The signal at 77.9 ppm was due to the solvent peak (cdcl 3).peaks at 110.12 and 115.04 ppm are assignable to the ch (cc and cd, respectively).the two adjacent signals at 118.85 and 122.89 ppm are attributable to the aromatic carbons, cf. Click on to reference a standard, such as tms, or a solvent peak (shortcut 'l'). Common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist gregory r. The chemical shifts were read and are presented in table 1. Why should the same species ($\ce{h2o}$) give rise to two different. A) residual h 2 o and hod in the d 2 o bottle;
The chemical shifts were read and are presented in table 1. Apart from cdcl3 other deuterated solvents in common use are. Under nmr spectrum scales vertical make sure vertical is chosen so you can pan in both. Why should the same species ($\ce{h2o}$) give rise to two different. Here, the chemical shifts of tms and the proton and deuterium chemical shifts of t …

A reference standard such as tetramethylsilane is commonly added (around 0.03%) to most commercially available solvent grades to serve as zero ppm chemical shift reference for nmr studies.
What is the reason behind it? Why does dmso show quintet in 1h nmr? Molecular weight, density (25 °c), melting point (mp), and boiling point (bp) are also given. Use the chart below to look up the coupling values — j hd and j cd (j cf) distance between multiplet peaks in hertz (hz) — and chemical shift delta values — ð h (mult) b and ð c (mult) b in parts per million (ppm) — of nmr solvents by name or cas number. Apart from cdcl3 other deuterated solvents in common use are. Under nmr spectrum scales vertical make sure vertical is chosen so you can pan in both. Nmr solvents deuterated solvents for nmr • nmr solvents • nmr reference standards • nmr tubes. Find the unknown molecule (the peaks at around 7.3ppm in the 1h nmr are due to the cdcl3 solvent, as are the three small peaks at around 77ppm in the 13c nmr) this problem has been solved! Günstige preise & mega auswahl für solvent. All of the compounds in table 1 were obtained as single isomers.
Gottlieb, 3 abraham nudelman, brian m. Nmr chemical shifts of trace impurities: All of the compounds in table 1 were obtained as single isomers. When the exchange rate between h0 and hdo is slow on the nmr timescale the water peak appears as two peaks, a singlet corresponding to h 20 and a 1:1:1 triplet cor responding to hdo. What is this peak due to and why the heck is it there? In chloroform solvent (cdcl 3), this corresponds to chcl 3, so a singlet signal is observed at 7.26 ppm.
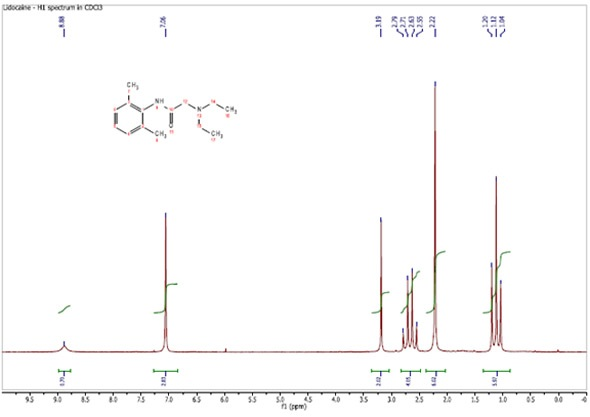
The standard for chemical shift is dilute tetramethylsilane (tms) in cdcl3, but many measurements are made relative to tms in other solvents, the proton resonance of the solvent peak or relative to the lock frequency.
A reference standard such as tetramethylsilane is commonly added (around 0.03%) to most commercially available solvent grades to serve as zero ppm chemical shift reference for nmr studies. Fig.5 indicates how a sample signal can be distinguished from a 13 c satellite by 13 c decoupling. Use the chart below to look up the coupling values — j hd and j cd (j cf) distance between multiplet peaks in hertz (hz) — and chemical shift delta values — ð h (mult) b and ð c (mult) b in parts per million (ppm) — of nmr solvents by name or cas number. Click on the arrow by to see the different options. Whenever you run a 13c spectrum in cdcl₃, you always get a triplet solvent peak at 77.5 ppm. However, whenever cdcl3 is used as an nmr solvent, a small singlet is always observed at 7.26 delta. A common solvent for dissolving compounds for 1h and 13c nmr spectroscopy is deuteriochloroform, dccl3. To avoid spectra dominated by the solvent signal, most 1 h nmr spectra are recorded in a deuterated solvent. What is this peak due to and why the heck is it there? Find the unknown molecule (the peaks at around 7.3ppm in the 1h nmr are due to the cdcl3 solvent, as are the three small peaks at around 77ppm in the 13c nmr) this problem has been solved! The chemical shifts were read and are presented in table 1. Sofort kostenlos und ohne anmeldung anfragen Günstige preise & mega auswahl für solvent. 3.8, gives rise to eight signals at 77.91, 110.12, 115.04, 118.85, 122.89, 132.25, 138.81, and 182.85 ppm. Mehr als 200.000 maschinen sofort verfügbar.
Use the chart below to look up the coupling values — j hd and j cd (j cf) distance between multiplet peaks in hertz (hz) — and chemical shift delta values — ð h (mult) b and ð c (mult) b in parts per million (ppm) — of nmr solvents by name or cas number cdcl3 nmr. Günstige preise & mega auswahl für solvent.
 Source: kovsky.net
Source: kovsky.net Günstige preise & mega auswahl für solvent.
 Source: jeolusa.s3.amazonaws.com
Source: jeolusa.s3.amazonaws.com Deuterated solvents, where 99+% of the protons are replaced with deuterium, are used as a solvent in nmr spectroscopy.
 Source: 4.bp.blogspot.com
Source: 4.bp.blogspot.com Use the chart below to look up the coupling values — j hd and j cd (j cf) distance between multiplet peaks in hertz (hz) — and chemical shift delta values — ð h (mult) b and ð c (mult) b in parts per million (ppm) — of nmr solvents by name or cas number.
To avoid spectra dominated by the solvent signal, most 1 h nmr spectra are recorded in a deuterated solvent.
 Source: employees.csbsju.edu
Source: employees.csbsju.edu 3.8, gives rise to eight signals at 77.91, 110.12, 115.04, 118.85, 122.89, 132.25, 138.81, and 182.85 ppm.
 Source: www.oc-praktikum.de
Source: www.oc-praktikum.de Common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist gregory r.
 Source: chem.libretexts.org
Source: chem.libretexts.org The chemical shifts were read and are presented in table 1.
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net It is also silent in the nmr and will not show peaks to interfere with the analysis of the compound of interest.
Why should the same species ($\ce{h2o}$) give rise to two different.
Mehr als 200.000 maschinen sofort verfügbar.
 Source: nmr.chem.ucsb.edu
Source: nmr.chem.ucsb.edu Why does the water peak appear at different chemical shift values (ppm) in different solvents in proton nmr spectra?
 Source: 4.bp.blogspot.com
Source: 4.bp.blogspot.com Common spectroscopic solvents are available commercially in different degrees of deuteration.
 Source: reader020.staticloud.net
Source: reader020.staticloud.net Why does dmso show quintet in 1h nmr?
 Source: ars.els-cdn.com
Source: ars.els-cdn.com The chemical shifts were read and are presented in table 1.
 Source: www.spectroscopyeurope.com
Source: www.spectroscopyeurope.com Günstige preise & mega auswahl für solvent.
Find the unknown molecule (the peaks at around 7.3ppm in the 1h nmr are due to the cdcl3 solvent, as are the three small peaks at around 77ppm in the 13c nmr) this problem has been solved!
.jpg) Source: d12oja0ew7x0i8.cloudfront.net
Source: d12oja0ew7x0i8.cloudfront.net Here, the chemical shifts of tms and the proton and deuterium chemical shifts of t …
 Source: www.qorganica.es
Source: www.qorganica.es Common laboratory solvents, organics, and gases in deuterated solvents relevant to the organometallic chemist gregory r.
 Source: static1.squarespace.com
Source: static1.squarespace.com Under nmr spectrum scales vertical make sure vertical is chosen so you can pan in both.
 Source: chem.ch.huji.ac.il
Source: chem.ch.huji.ac.il What is this peak due to and why the heck is it there?
3.8, gives rise to eight signals at 77.91, 110.12, 115.04, 118.85, 122.89, 132.25, 138.81, and 182.85 ppm.
 Source: chem.ch.huji.ac.il
Source: chem.ch.huji.ac.il What is this peak due to and why the heck is it there?
 Source: d2vlcm61l7u1fs.cloudfront.net
Source: d2vlcm61l7u1fs.cloudfront.net A common solvent for dissolving compounds for 1h and 13c nmr spectroscopy is deuteriochloroform, dccl3.
 Source: www.chemguide.co.uk
Source: www.chemguide.co.uk Pure deuterated dmso shows no peaks in 1 h nmr spectroscopy and as a result is commonly used as an nmr solvent.
 Source: 3.bp.blogspot.com
Source: 3.bp.blogspot.com Jan 9, 2015 the deuterium in the cdcl₃ splits the 13c signal into a triplet.
 Source: 2.bp.blogspot.com
Source: 2.bp.blogspot.com Find the unknown molecule (the peaks at around 7.3ppm in the 1h nmr are due to the cdcl3 solvent, as are the three small peaks at around 77ppm in the 13c nmr) this problem has been solved!
 Source: chem.ch.huji.ac.il
Source: chem.ch.huji.ac.il This is a standard reference point with the signal set exactly at 0 ppm and y ou can ignore it when analyzing an nmr spectrum.
 Source: chem.libretexts.org
Source: chem.libretexts.org The chemical shifts were read and are presented in table 1.
Günstige preise & mega auswahl für solvent.
 Source: www.oc-praktikum.de
Source: www.oc-praktikum.de What is the reason behind it?
 Source: ars.els-cdn.com
Source: ars.els-cdn.com The chemical shifts were read and are presented in table 1.
Posting Komentar untuk "Cdcl3 Nmr Solvent Peak"